GMP, 100mM disodium salt solution
GMP, 100mM disodium salt solution - M (Molecular Biology) / 1mL / centrifuge tube is backordered and will ship as soon as it is back in stock.
Couldn't load pickup availability
Shipping notes
Shipping notes
Details
Details
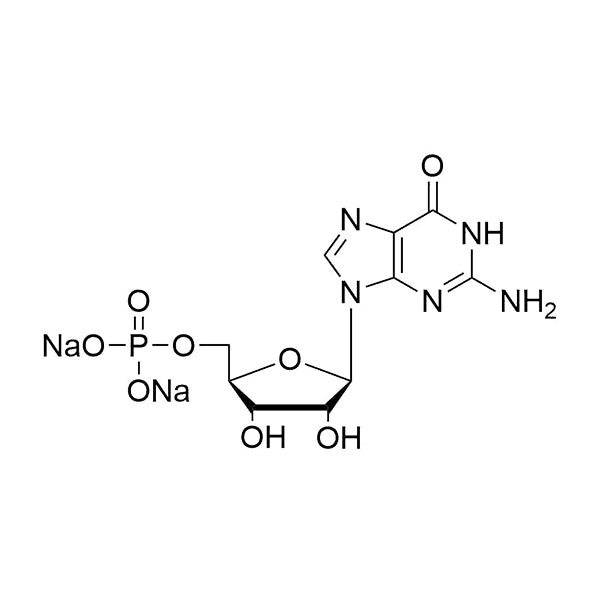
Guanosine-5’-monophosphate (GMP) is a naturally occurring ribonucleotide composed of guanine, ribose, and a single 5’-phosphate group.
GMP serves as a key biochemical building block in RNA metabolism, enzymatic reactions, and biosynthesis studies.
This 100 mM disodium salt solution provides GMP in a highly soluble, ready-to-use form suitable for both routine laboratory applications and high-consistency manufacturing environments.
Applications
Enzymatic & Biochemical Research
Acts as a substrate or regulator in studies involving nucleotide biosynthesis, RNA polymerases, phosphotransfer enzymes, and signal-transduction pathways.[1]
Reference Standards & Controls
Serves as a quantitative standard for HPLC, LC–MS, enzyme kinetics, and calibration of nucleotide quantification assays.[2]
Features and Benefits
Reference
Specifications
Specifications
-
Catalog No.R21312
-
SMILESO=C1C(N=CN2[C@H]3[C@H](O)[C@H](O)[C@@H](COP(O[Na])(O[Na])=O)O3)=C2N=C(N)N1
-
Molecular FormulaC10H12N5Na2O8P
-
Molecular Weight407.19
-
AppearanceClear colorless solution
-
PurityHPLC≥98%
-
Storage Condition-20℃
-
Bacterial Endotoxin Test<1 EU/ml
-
Quality ControlThis preparation is free of DNase and RNase contamination.
Documentation
Documentation
Guanosine-5’-monophosphate (GMP) is a naturally occurring ribonucleotide composed of guanine, ribose, and a single 5’-phosphate group.
GMP serves as a key biochemical building block in RNA metabolism, enzymatic reactions, and biosynthesis studies.
This 100 mM disodium salt solution provides GMP in a highly soluble, ready-to-use form suitable for both routine laboratory applications and high-consistency manufacturing environments.
Applications
Enzymatic & Biochemical Research
Acts as a substrate or regulator in studies involving nucleotide biosynthesis, RNA polymerases, phosphotransfer enzymes, and signal-transduction pathways.[1]
Reference Standards & Controls
Serves as a quantitative standard for HPLC, LC–MS, enzyme kinetics, and calibration of nucleotide quantification assays.[2]
Features and Benefits
Reference
-
Catalog No.R21312
-
SMILESO=C1C(N=CN2[C@H]3[C@H](O)[C@H](O)[C@@H](COP(O[Na])(O[Na])=O)O3)=C2N=C(N)N1
-
Molecular FormulaC10H12N5Na2O8P
-
Molecular Weight407.19
-
AppearanceClear colorless solution
-
PurityHPLC≥98%
-
Storage Condition-20℃
-
Bacterial Endotoxin Test<1 EU/ml
-
Quality ControlThis preparation is free of DNase and RNase contamination.
Why choose Hongene?
Trusted Partner in Nucleic Acid
Integrated Supply & Commercial Scale
With 26+ years of expertise, we control a secure supply chain for RNA raw materials and provide reliable GMP-grade oligo synthesis from research to commercial kilogram-scale production.
Proprietary Technology & IP
Our proprietary Chemoenzymatic Ligation Platform combines chemical andenzymatic methods, enabling high-putity, cost-effective, and large-scale production of RNA-based therapeutics.
Rigorous Quality
We implement multiple stringent QC steps, maintain ISO certifications, and ensure >99% batch-to-batch consistency, reducing scale-up and PPQ risks.
Manufacturing Scalability
Hongene operates 1.67 million sq. ft Oligonucleotide Manufacturing Facility, with advanced equipments including multiple OligoPilot™ and OligoProcess™ synthesizers (10-1800 mmol). 48 flexible production lines enable one-stop seamless scaling-up of API production from gram-level to tons and acheive high purity of 98%, meeting NMPA, FDA, and EMA standards.
Global Business Network
Our products and services reach over 40 countries and regions, supporting around 3,000 clients worldwide.